Mesa Labs Bacillus smithii Spore Suspension for Sterilization Efficacy Testing (Model MESSSME7-0)
Spore Suspension Bacillus smithii 51232
Reliable Biological Indicator for Sterilization Monitoring
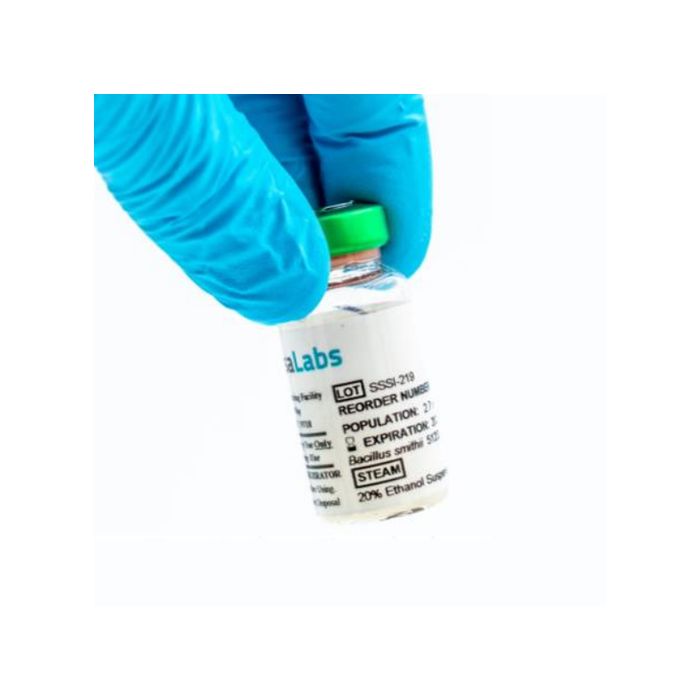
Spore Suspension Bacillus smithii 51232 contains highly resistant spores in a liquid suspension. Each 10 mL Wheaton vial includes a rubber stopper and a colored aluminum crimp seal for easy species identification. This calibrated spore suspension ensures consistent performance for sterilization validation and routine monitoring.
Applications
This suspension is intended for direct inoculation onto products or substrates to verify low-temperature steam sterilization processes. It is widely used in pharmaceutical, medical device, and laboratory sterilization testing, where precise validation of sterilization efficacy is required.
Packaging Details
Each 10 mL vial is sealed with a green aluminum crimp and individually packaged in a clear plastic bag. This packaging preserves sterility and simplifies handling during testing.
Labeling Information
Vials are clearly labeled with the species name, storage conditions, lot number, and spore population count, facilitating traceability and compliance with standard protocols.
Additional spore products tailored to specific sterilization are available such as the Spore Suspension Bacillus subtilis 5230, engineered for low‑temperature steam applications with heat‑sensitive product profiles, or the Apex Spore Suspension Geobacillus stearothermophilus, optimized for high‑temperature steam sterilization monitoring and media validation. For dry heat and ethylene oxide control points, the Spore Suspension Bacillus atrophaeus 9372 provides a reliable biological challenge agent to support regulatory compliance across complex industrial sterilization cycles.
Q&A
What is the recommended storage for Bacillus smithii spore suspensions?
Store vials at 2–8 °C to maintain spore viability. Avoid freezing or prolonged exposure to high temperatures.
How should I use this spore suspension for sterilization validation?
Directly inoculate the target product or substrate according to your low-temperature steam sterilization protocol, then incubate and monitor for growth to assess sterilization effectiveness.
Can this suspension be used for different sterilization processes?
It is specifically designed for low-temperature steam sterilization. For other sterilization methods (e.g., dry heat, ethylene oxide), choose a suspension validated for that process.
Is the spore population consistent across vials?
Yes, each lot is calibrated to provide a consistent spore count for reliable, repeatable results.
Where can I find similar spore suspensions?
- Spore Suspension Geobacillus stearothermophilus 51221
- Spore Suspension Bacillus atrophaeus 51226
- Spore Suspension Geobacillus stearothermophilus 51222
- Spore Suspension Bacillus subtilis 51223
| Organism | Bacillus smithii 512321 |
| Spore Count Options (per 0.1mL) | ≥1.0 x 107 model: SSSME/7 |
| D-Value | No standard range |
| Z-Value | NA |
| *Reference D-Value from ampoules containing Water For Injection | |
| Diluent | 20% ethanol (EtOH) |
| Storage | 2˚C to 8˚C |
| Incubation | Temperature: 48-52˚C Media: SCDB |
| Shelf-Life | 24 months from date of manufacture |
| Nominal Dimensions | Volume: 10mL |
| Price | $USD 2,956.00 |
|---|
Need larger scale equipment for a bigger project?
Our parent site geneq.com works by quote requests, so we can offer a unique price per any combination of items you choose, and the best shipping company/route for weighty items. We distribute the biggest names in the industry since 1972!